What is "Science" ?
What is a hypothesis and how do you make one ?
I think science is a hands-on experiment and a observing subject.
Update : I think science is a subject where you can be curious, take notes, do experiments and learn different things.
I think a hypothesis is a "guess" which is based on evidence upon the subject.
Update : I think science is a subject where you can be curious, take notes, do experiments and learn different things.
I think a hypothesis is a "guess" which is based on evidence upon the subject.
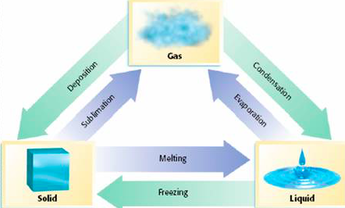
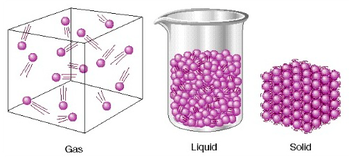
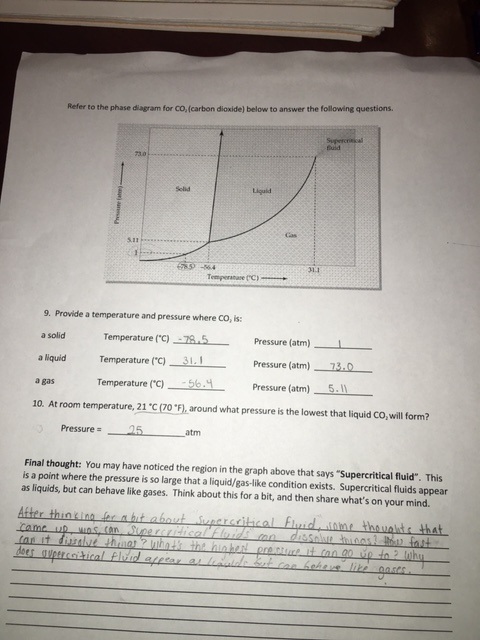
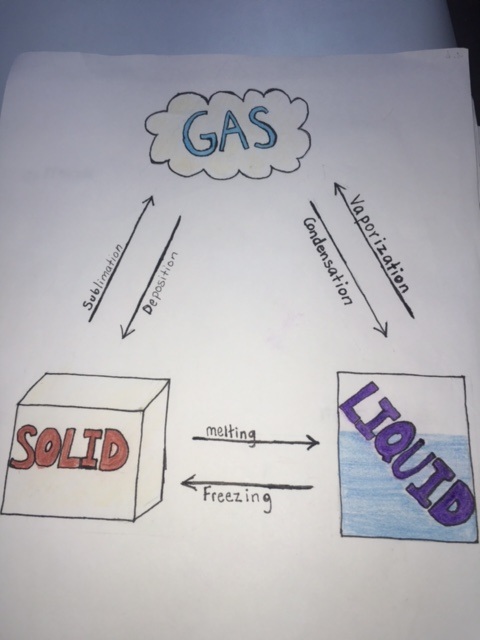
Phases of Matter

There are 3 phases of matter which are Gas, Liquid, and Solid. Gas has atoms bouncing off each other, liquid has lose packed atoms and solid has tightly packed atoms. For our experiment our teacher took dry ice from the freezer and put in a room temperature area. He put some dry ice into a tube and sealed it. It turned out to be a gas instead of a liquid because there wasn't enough pressure to be form into liquid. Once there was enough pressure it turned to a liquid and once he released the pressure of the tube the liquid coming out turned out to be snow.
|
|
In chemistry we did an experiment which involved magnesium, citric acid and water. We mixed these components together and it made hydrogen. A combustion needs 3 things in order to happen ; a fuel, an oxidizer, and a ignition stimulus.
What do you think happened when we burned our hydrogen balloons?
I think what happened to the balloon's was that since there was hydrogen in it, the hydrogen had a reaction to the fire which caused the loud popping noise. There were so many atoms bumping into each other that made the balloon go crazy.
Update: In this experiment, we mixed water, citric acid and magnesium, filled the balloon with a unstable gas. All atoms have 3 sub-atomic particles called protons, neutrons and electrons. By being able to burn the balloon it made the atoms go all crazy, because it was not stable so a combustion occurred when we put the fire on the balloon |
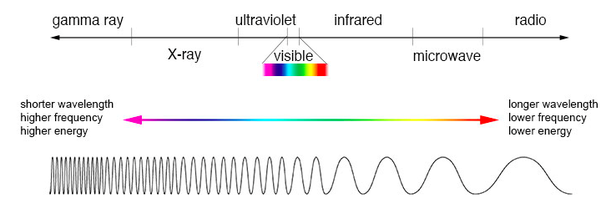
What is light ?
Light is energy, that allows us to see, in different colors and everything around us. Light is made up of wavelengths which are shown on the Electromagnetic Spectrum.
Properties Of Matter

In class we learned the properties of matter. We did a experiment which included 4 candles, but we had to cut them in half, 2 pieces of balsa wood and a trash bag. My group and I decided to put the to balsa wood pieces to make an X and put 4 candles on each balsa wood and tapped all four sides of the trash bag to the wood. As we lit our candles the bag began to expand and little by little the bag began to fly because of all the air molecules bouncing off each other. When there's higher energy the faster it moves. When there is less energy the less it moves. Pressure is the sum of the force caused by the molecules bouncing off each others. Temperatures affects pressure, pressure affects volume.
Disappearing Ink
|
|
In the disappering ink lab we measured .1g of phenophthalien. Added 100ml of ethanol and then added 100ml of DL H2O and we dropped 3m NaOH until it was dark red. Then we shook it which changed the pH level to neutral bringing the liquid back to the original clear form.
|
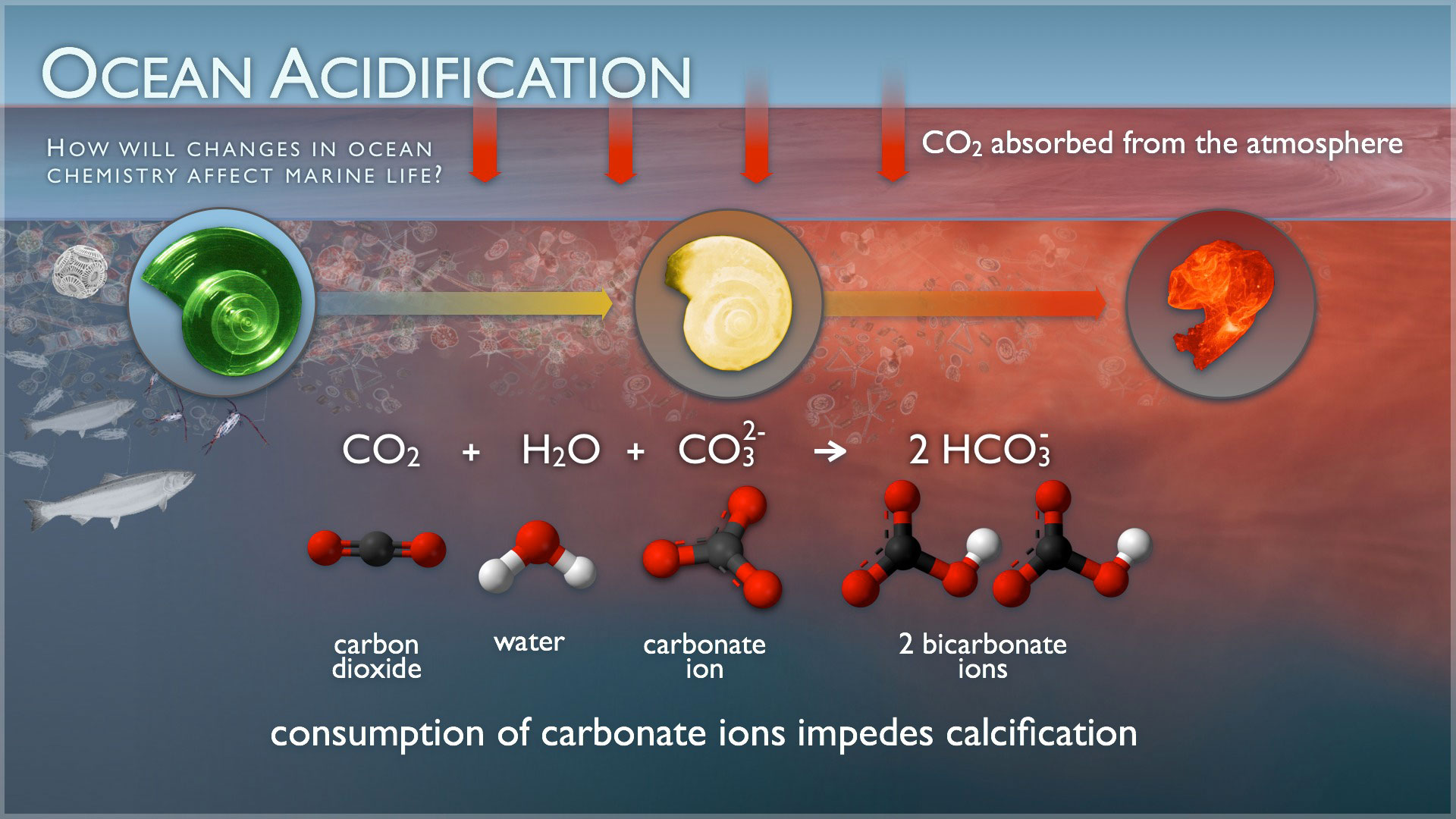
Ocean Acidification
|
Ocean Acidification is measured by a pH scale. On the pH scale each step is a factor of ten. 0 would be highly acidic and 14 is highly alkaline. If you increase acidicy the pH is dropping. If you increase alkalinity the pH is going up. pH is a measure of potential. In 1751-1990's the pH decreased from 8.25-8.14 which doesnt seem like a lot but in reality it is because from 6 to 5 the acidic level is decreasing by 10 times than before. CO2 is in the atmosphere and it gets into the ocean. When carbon dioxide is absorbed by seawater it causes a chemical reaction to occur. By that happening it causes the seawater to become more acidic and causes carbonate ions to be less abundant. Ocean acidification is affecting the entire world's ocean, waterways and ravines.
|